Beyond the Box-and-Arrow
Timoteo Almeida, MD, PhD | Department of Neurological Surgery
Orientation
This reading is built around a single clinical observation that most trainees — and most textbooks — never quite resolve: STN DBS routinely cuts levodopa equivalent daily dose (LEDD) by 40–50%. GPi DBS rarely cuts it by more than 20%. Yet motor improvement is roughly equivalent between the two targets, and the Albin-DeLong circuit diagram we all grew up with predicts nothing of the kind.
Learning to sit with that apparent paradox is a useful apprenticeship. The resolution is not a single mechanism; it is three overlapping stories — a clinical-management story, a pathophysiological story, and a modern network/sweet-spot story. Each layer corrects the previous one without replacing it. By the end of this reading, you should be able to explain to a patient why their neurologist is tapering their Sinemet after STN DBS and leaving it largely alone after GPi DBS, and you should be able to predict which of your future patients will taper well and which will not.
THE MECHANISTIC DISCONNECT
1. The Apparent Paradox
The classical Albin-DeLong model describes two parallel striatofugal pathways. The direct pathway (striatum → GPi/SNr) facilitates movement; the indirect pathway (striatum → GPe → STN → GPi/SNr) inhibits it. Nigral dopamine loss disinhibits the indirect pathway, which drives the STN into a hyperactive, abnormally bursting state, which in turn overdrives the GPi, which in turn over-inhibits the thalamus. Bradykinesia and rigidity follow.
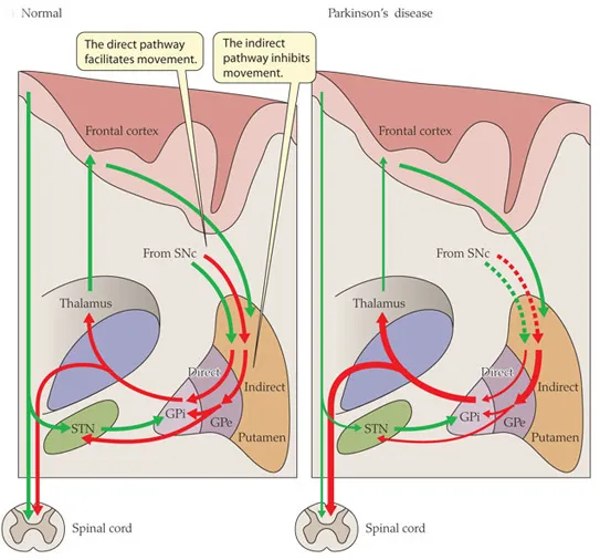
Figure 1. The classical Albin-DeLong rate model. Left: normal circuit. Right: Parkinson disease, with exaggerated indirect-pathway drive (thick red arrows) producing STN hyperactivity and excess GPi output. Read literally, the schematic predicts that modulating either STN or GPi should yield equivalent downstream effects — an intuition the clinical data dismantle.
Read literally, this schematic tells you that STN and GPi are two points on a serial wire. Silencing either node should produce a comparable downstream effect. The clinical trial data partly support that intuition — motor scores improve similarly at both targets — but then diverge sharply when you look at medication: in CSP 468, NSTAPS, and essentially every comparative meta-analysis, STN DBS is associated with a substantially greater LEDD reduction (40–50%), while GPi DBS typically reduces LEDD by 0–20%. The schematic does not predict this.
2. First Layer — The Clinical-Practice Answer
The cleanest, most immediately useful framework was articulated by Follett in 2004 and has held up: GPi DBS has a direct anti-dyskinetic effect; STN DBS does not (or only weakly so). This asymmetry drives almost everything downstream about medication management.
After GPi implantation, stimulation itself suppresses dyskinesia regardless of how much levodopa the patient is taking. The neurologist can leave the LEDD essentially unchanged and still get excellent dyskinesia control. There is no mechanical pressure to taper.
After STN implantation, the opposite is true: if you do not reduce levodopa, the patient develops or worsens stim-unmasked dyskinesia. Programming and medication titration are therefore coupled — the team is practically forced to taper the LEDD to get the motor benefit without the chorea. A large fraction of the LEDD-reduction gap between targets is a management artifact, not a distinct pharmacodynamic effect.
A useful shorthand is "ceiling versus floor": GPi raises the ceiling on how much levodopa a patient can tolerate without dyskinesia; STN raises the floor of motor function so levodopa becomes less necessary. The heuristic is memorable, but be honest about what it hides: it treats the pharmacology as purely a clinical-management story and says nothing about why STN stimulation substitutes for some of what levodopa was doing. For that we need the pathophysiology.
A practical corollary: work from the Xuanwu group has shown that STN DBS can suppress levodopa-induced on-dyskinesia without any medication reduction, particularly when stimulation is delivered dorsal to the nucleus. So "STN has no direct anti-dyskinetic effect" is too strong — the correct statement is that GPi’s direct effect is much larger and reliable, while STN’s direct effect is real but smaller, contact-dependent, and usually overwhelmed in practice by the need to taper anyway.
3. Second Layer — Where Dopamine Actually Acts
Return to the diagram, and ask a more careful question: where does dopamine physiologically act? The answer is the striatum, not the GPi.
Nigrostriatal dopamine tunes both pathways at the input side of the basal ganglia circuit — D1 drive facilitates the direct pathway, D2 drive suppresses the indirect pathway. Loss of nigral dopamine propagates down the indirect pathway as pathological STN output. That pathological output is what drives the symptomatic over-inhibition of the thalamus. This is the physiological site of levodopa’s action.
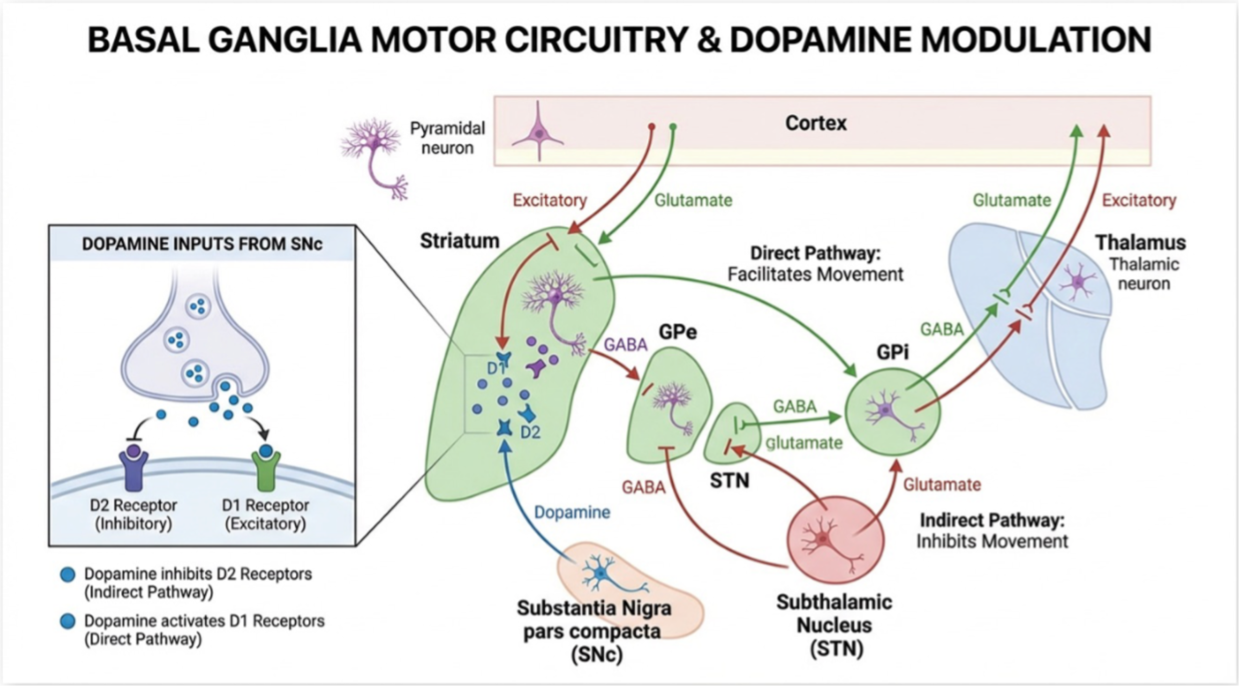
Figure 2. Where dopamine acts. Nigrostriatal dopamine modulates both pathways at the striatal input stage via D1 (excitatory, direct-pathway facilitation) and D2 (inhibitory, indirect-pathway suppression) receptors. This is the physiological site of levodopa’s action — not the GPi. Understanding this is the key to understanding why STN DBS and GPi DBS have such different pharmacological consequences.
Now look at where each DBS target sits relative to that physiology. STN DBS sits at the node dopamine was normally regulating through the indirect pathway. Suppressing or replacing the pathological STN signal effectively substitutes for part of what dopaminergic tone was doing — there is a real physiological overlap between what levodopa does and what STN stimulation does, even though the proximate mechanism differs (D2 receptor action in the striatum versus electrical modulation of STN neurons and their afferents/efferents). That overlap is the reason patients can take less drug and still function.
GPi DBS sits one station further downstream, at the output of the basal ganglia. It masks or overrides the final common output regardless of what is feeding in. The upstream pathological drive is still there; you have just silenced its expression at the gate. There is no substitution for dopaminergic function — the striatum still wants its dopamine — and so there is no physiological room to cut the drug.
A third mechanism supports this framing. STN neurons send collaterals to the substantia nigra pars compacta, and PET imaging with the VMAT2 tracer [¹¹C]-DTBZ has shown changes in striatal dopamine-related binding after STN DBS that are consistent with enhanced release from surviving nigral neurons. GPi DBS, lacking this direct feedback onto the SNc, cannot do this. It is a small mechanism quantitatively, but it is a real dopamine-sparing effect that only STN stimulation possesses.
4. Third Layer — The Modern Network View
The Albin-DeLong model was built on firing rates. The modern view is built on firing patterns — specifically, pathological synchrony in the beta band (13–30 Hz) across the cortico-basal ganglia-thalamic loop. In PD, beta oscillations dominate the STN local field potential, and their amplitude scales with bradykinesia and rigidity. Dopaminergic replacement suppresses them. High-frequency STN DBS suppresses them too. The convergence is not accidental.
The hyperdirect pathway. Oswal and colleagues (2021) used simultaneous MEG and STN-LFP recordings with structural tractography to show that high-beta (21–30 Hz) coherence between the supplementary motor area and STN correlates directly with hyperdirect pathway fiber density — and that cortex drives STN at these frequencies, not the other way around. The sweet spot for STN DBS — the dorsolateral border of the nucleus and the adjacent white matter — is exactly where these hyperdirect fibers enter. Akram et al. (2017) mapped the cortical fingerprint of effective STN stimulation and showed that tractographic connectivity to M1 predicts tremor improvement, to SMA predicts bradykinesia improvement, and to both SMA and prefrontal cortex predicts rigidity improvement. The best current mechanistic account is that STN DBS works in large part by antidromic modulation of these cortico-STN fibers, disrupting the pathological cortex-to-STN beta coupling at its source.
ERNA and the indirect pathway. Evoked resonant neural activity — the stimulation-locked high-frequency oscillation that appears in the STN during DBS — is now understood (Steiner et al., 2024) as a signature of DBS-mediated engagement of the GPe-STN indirect-pathway loop. ERNA localization predicts motor outcome, and it is engaged by both STN and GPi stimulation. This is probably part of why GPi DBS works at all despite being "downstream" of dopamine’s site of action, and why both targets produce similar motor improvement despite their different relationships to the drug.
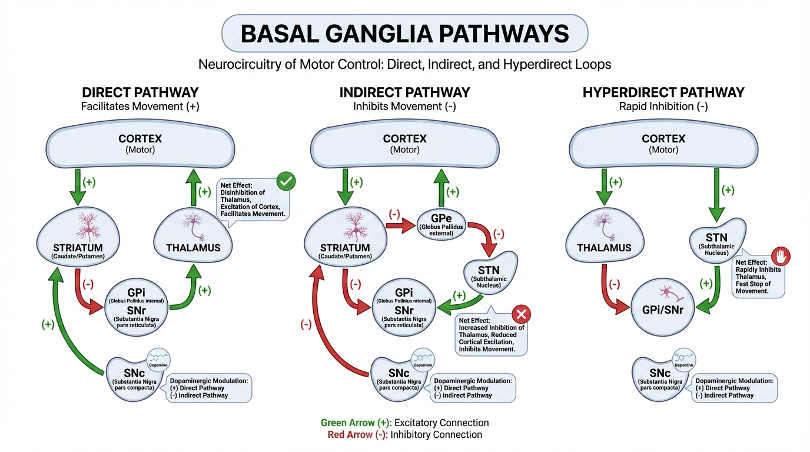
Figure 3. The three cortical-to-basal-ganglia pathways. The hyperdirect pathway (right) is the piece missing from the classical Albin-DeLong schematic in Figure 1. Fibers from primary motor and pre-motor cortex — notably SMA — descend through the internal capsule and terminate directly on the STN, bypassing the striatum. This pathway is the anatomical substrate for rapid cortical pacing of the STN and for the pathological beta oscillations that characterize the parkinsonian state.
Two practical implications. First, "STN DBS mimics levodopa" is a useful teaching line but mechanistically imprecise — they converge on the same physiological signature (suppression of pathological beta synchrony) via different molecular mechanisms. Second, GPi DBS does not achieve the same cortex-STN beta suppression via the hyperdirect pathway, which is part of why it cannot substitute for the drug.
CLINICAL APPLICATION & TARGET SELECTION
5. The Dissociation — Good Lead Placement Does Not Equal Big LEDD Reduction
This is the advanced nugget, and it is the one most clinically useful to carry into the OR and the programming clinic. Motor improvement and LEDD reduction are not the same outcome, and they do not scale together. They have partially different predictors.
The Dembek 2019 Annals of Neurology paper quantified how much motor improvement comes from hitting the correct anatomical target. Overlap of the volume of tissue activated (VTA) with the probabilistic sweet spot — located at the dorsolateral border of the STN, extending into the adjacent white matter — explained R² = 37% of the variance in motor improvement, more than triple what was explained by raw STN coverage (9%) or sensorimotor STN coverage (10%). That sweet spot is approximately the hyperdirect pathway entry zone. In short: lead placement matters enormously for motor outcome, and centroid-of-STN is a weaker predictor than sweet-spot overlap.
Now the Benoit-Marand 2026 mediation analysis: 144 patients, Boruta feature selection, structural equation model to separate direct effects on LEDD reduction from motor-mediated effects. Mean LEDD reduction was 41.7%, mean motor improvement 48.6%. Three predictors were directly associated with lower LEDD reduction, independent of motor improvement: lower baseline LEDD (the floor-effect), greater axial impairment, and — importantly — higher total volume of tissue activated. Sensorimotor STN overlap was positively associated with motor improvement but not with LEDD reduction.
This is the mechanistic argument for directional leads, for image-guided programming, and for tractography-informed contact selection. It is also the argument against promising a patient any specific LEDD reduction preoperatively: a patient with heavy axial disease or relatively low baseline LEDD will not drop their meds by 50% no matter how good your lead placement is, because the biology will not allow it. That is not a failure of technique — it is the separate axis asserting itself.
6. What the Comparative Trials Actually Show
Four studies form the spine of the evidence base. Read at least the first three in full before lecture.
Limousin 1995 (Lancet). The founding three-patient series of bilateral STN DBS for advanced PD. Activities of daily living improved 58–88%; motor scores 42–84%. This is where the field starts, and the paper is short enough to read in fifteen minutes.
CSP 468 / Follett 2010 (NEJM). 299 patients, VA-NINDS multicenter, randomized STN versus GPi. Primary outcome — off-medication, on-stimulation UPDRS-III — did not differ significantly between targets. Patients in the STN arm required significantly lower dopaminergic doses (P=0.02); STN patients had a greater decline in one component of processing speed (visuomotor, P=0.03); depression worsened after STN and improved after GPi (P=0.02). This is the trial that made "target selection should be individualized" an evidence-based statement rather than a prejudice.
NSTAPS / Odekerken 2013 (Lancet Neurology). Dutch multicenter RCT, 128 patients. Hypothesis: GPi stimulation would produce greater functional improvement than STN because of fewer cognitive/mood complications. The data contradicted the hypothesis: STN was better on off-period motor improvement and disability, dyskinesia reduction was larger with GPi (57% vs 21% reduction on blinded CDRS at 12 months), and LEDD reduction was significantly greater with STN. Three-year follow-up (Odekerken 2016) confirmed the motor advantage and the absence of a cognitive safety penalty for STN.
CSP 468F / Ostrem 2025 (Frontiers in Neurology). Ten-year extension of CSP 468. Motor improvement was sustained in both arms. The LEDD reduction gap between targets narrows considerably at very long follow-up — both groups ended up with significant medication reduction and no difference in time trend (P=0.70). Useful to close the loop with, because it reminds trainees that "STN spares meds more than GPi" is largely a short-to-intermediate-term phenomenon; disease progression drives both groups toward similar LEDD trajectories over a decade.
7. Target Selection Framework
Given the two targets are equivalent for motor improvement, selection is driven almost entirely by the patient’s specific profile of symptoms, side effects, cognitive reserve, and treatment goals. A pragmatic framework for lecture:
Favors STN: younger patient with intact cognition and mood; medication burden is the problem (impulse control disorder, dopamine dysregulation syndrome, dopa-induced nausea, prominent medication-induced hallucinations); battery longevity or economic considerations favor the lower stimulation amplitudes typical of STN; severe off-period bradykinesia/rigidity; need for more off-period motor improvement.
Favors GPi: dyskinesia-dominant disease with heavy reliance on levodopa that you do not want to taper (e.g., patient requires the drug for cognitive/mood stability); baseline cognitive vulnerability or mild cognitive impairment; depression or emotional lability at baseline; more advanced/older patient; programming flexibility is important; unilateral implantation is anticipated; surgeon or team is earlier on the learning curve (GPi is more forgiving).
Both targets fail: axial symptoms (gait freezing, postural instability, speech) and non-levodopa-responsive symptoms. The levodopa challenge is your most reliable predictor of DBS motor outcome — if a symptom does not respond to dopamine, it will not reliably respond to stimulation at either target. Setting this expectation preoperatively is half the battle.
8. Pearls for the Operating Surgeon
The target is the dorsolateral STN and the adjacent white matter, not the center of the nucleus. "Nice lead through the middle of STN on post-op MRI" is a weaker predictor of outcome than "VTA overlapping the sweet spot and the hyperdirect pathway entry zone." Plan accordingly.
A smaller, focused VTA beats a larger one. Bigger stimulation fields recruit non-motor STN territory and its side effects, narrowing the therapeutic window and capping how aggressively the neurologist can cut meds. Directional leads earn their cost here.
Motor outcome and LEDD reduction are separate axes with different predictors. Do not promise a patient a specific LEDD drop preoperatively. Promise motor improvement contingent on levodopa responsiveness, and discuss medication management as a separate question that depends on their baseline profile and how the team co-manages stim and drug.
The forced taper is therapeutic, not incidental. For patients with dopamine dysregulation syndrome, severe ICDs, or dopamine-induced hallucinations, STN DBS is the right answer specifically because the mandatory medication reduction is itself the treatment for the thing that was disabling them. Say this explicitly in the clinic.
Read Limousin 1995 before lecture. The paper is four pages, reports three patients, and is the reason we have a field. Trainees should know where their work comes from.
